Evolution of interface and morphology of soft nanolayer on Si
The interfacial stability or instability can strongly influence the growth of nanolayers of soft materials [e.g. Langmuir-Blodgett (LB) film of amphiphiles] on semiconductor substrates and accordingly, interesting nanostructures can evolve with time. This stability or instability at ambient condition depends on substrate surface passivation. Surface passivation modifies the polar-nonpolar nature of the surface and accordingly, hydrophilic-hydrophobic nature of the surface can be tuned. This essentially controls the growth and stability of the LB films or can control the formation of interesting metal-organic nanostructures. When nanolayer is made of metal-organic molecules, which have hydrophilic headgroups and hydrophobic tails, then hydrophilic, hydrophobic or coexisting nature of the terminated Si surfaces control the structure of the LB films. Presence of both hydrophilic and hydrophobic regions creates interesting nanostructures to release interfacial stress either through buckling or collapse of molecules. The hydrophilic-hydrophobic (polar-nonpolar) nature of the known surface in molecular level or transition of nature of surface (from hydrophobic to hydrophilic) is possible to find out by knowing the structure of LB film on it.
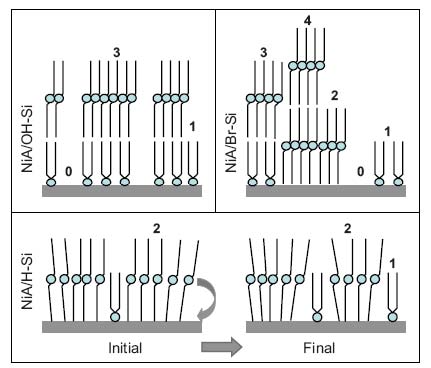 Growth of LB films of NiA on differently terminated (OH-, H-, or Br-terminated) Si(001) substrates and their structural evolution with time have been investigated by x-ray reflectivity technique and complemented by atomic force microscopy. Stable and strongly attached asymmetric monolayer (AML) of NiA is found to grow on freshly prepared oxide-covered Si substrate while unstable and weakly attached symmetric monolayer (SML) of NiA grows on H-terminated Si substrate, corresponding to stable hydrophilic and unstable hydrophobic natures of the substrates, respectively. The structure of LB film on Br-terminated Si substrate, however, shows intermediate behavior, namely, both AML and SML are present on the substrate, indicative of coexisting (hydrophilic and hydrophobic) nature of this terminated surface. Such coexisting nature of the substrate shows unusual growth behavior of LB films: (i) hydrophilic and hydrophobic attachments of NiA molecules in single up stroke of deposition and (ii) growth of few ring-shaped largeheights islands in subsequent deposition. These probably occur due to the presence of substrate-induced perturbation in the Langmuir monolayer and release of initially accumulated strain in the film structures near hydrophilic/hydrophobic interface, respectively, and provide the possibility to grow desired structures (AML or SML) of LB films by passivation-selective surface engineering [published in Phys. Rev. B 81, 045404 (2010)].
Growth of LB films of NiA on differently terminated (OH-, H-, or Br-terminated) Si(001) substrates and their structural evolution with time have been investigated by x-ray reflectivity technique and complemented by atomic force microscopy. Stable and strongly attached asymmetric monolayer (AML) of NiA is found to grow on freshly prepared oxide-covered Si substrate while unstable and weakly attached symmetric monolayer (SML) of NiA grows on H-terminated Si substrate, corresponding to stable hydrophilic and unstable hydrophobic natures of the substrates, respectively. The structure of LB film on Br-terminated Si substrate, however, shows intermediate behavior, namely, both AML and SML are present on the substrate, indicative of coexisting (hydrophilic and hydrophobic) nature of this terminated surface. Such coexisting nature of the substrate shows unusual growth behavior of LB films: (i) hydrophilic and hydrophobic attachments of NiA molecules in single up stroke of deposition and (ii) growth of few ring-shaped largeheights islands in subsequent deposition. These probably occur due to the presence of substrate-induced perturbation in the Langmuir monolayer and release of initially accumulated strain in the film structures near hydrophilic/hydrophobic interface, respectively, and provide the possibility to grow desired structures (AML or SML) of LB films by passivation-selective surface engineering [published in Phys. Rev. B 81, 045404 (2010)].
 Systematic investigation was carried out for CdA LB films on differently terminated Si to find out the role of metal ions on the growth different structure and its stability and the mechanism behind different growth and stability. For that CdA LB films were deposited on differently terminated Si(001). Also CdA LB films on H-passivated Si substrates were deposited in different strokes by keeping the substrates in subphase water having different pH with or without Cd metal ions and for different time. The structure of the LB films and its evolution with time were monitored using XR and AFM techniques. XR data were collected as a function of time. AFM images were collected in tapping mode to minimize the tip induced damage of soft materials, few days after deposition. The hydrophilic or hydrophobic nature of the Si surface treated differently was verified using home built contact angle measurement setup. XPS measurements were carried out to know the chemical nature of the Si surface after treatment and after LB film deposition [published in Eur. Phys. J. E 35, 79 (2012)].
Systematic investigation was carried out for CdA LB films on differently terminated Si to find out the role of metal ions on the growth different structure and its stability and the mechanism behind different growth and stability. For that CdA LB films were deposited on differently terminated Si(001). Also CdA LB films on H-passivated Si substrates were deposited in different strokes by keeping the substrates in subphase water having different pH with or without Cd metal ions and for different time. The structure of the LB films and its evolution with time were monitored using XR and AFM techniques. XR data were collected as a function of time. AFM images were collected in tapping mode to minimize the tip induced damage of soft materials, few days after deposition. The hydrophilic or hydrophobic nature of the Si surface treated differently was verified using home built contact angle measurement setup. XPS measurements were carried out to know the chemical nature of the Si surface after treatment and after LB film deposition [published in Eur. Phys. J. E 35, 79 (2012)].
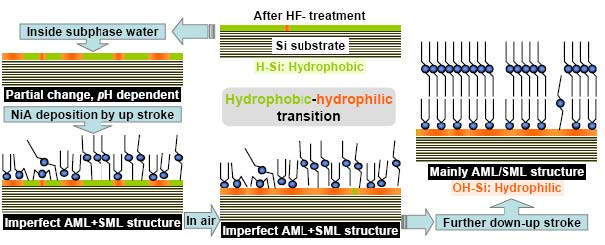 HF-treated Si surface, which is hydrophobic in nature and quite stable in air, transformed partially or completely to hydrophilic surface, inside subphase water or during NiA LB film growth by up-down-up strokes of deposition and in ‘high pH’ condition. This transition essentially takes place by conversion of H-terminated Si surface to OH-terminated one, through oxide formation. Although the amount of oxygen inside subphase water is less compared to that in air, the oxide formation inside subphase water is more, which takes place by chemical reaction in presence of highly electronegative OH- ions. As the pH of the subphase water increases, the amount of OH- ions increases and so the amount of oxide coverage. Also, as the duration for which the substrate was inside subphase water increases, the chances of chemical reaction increases and so the amount of oxide coverage, but never complete or full. The attachment of Ni head groups on the oxide portion of the partially transformed Si substrate by deposition of LB film, essentially weaken the nearby Si-Si bonding, which then readily react with oxygen to form oxides. It is observed that depending upon the amount of attached Ni head groups and its distribution on the Si surface, it is possible to have complete oxide coverage through such reaction in air. The signature of which, from structural point of view, is only visible after subsequent deposition, i.e. after reorganization of molecules under subphase water [published in Chem. Phys. Lett. 500, 90 (2010)].
HF-treated Si surface, which is hydrophobic in nature and quite stable in air, transformed partially or completely to hydrophilic surface, inside subphase water or during NiA LB film growth by up-down-up strokes of deposition and in ‘high pH’ condition. This transition essentially takes place by conversion of H-terminated Si surface to OH-terminated one, through oxide formation. Although the amount of oxygen inside subphase water is less compared to that in air, the oxide formation inside subphase water is more, which takes place by chemical reaction in presence of highly electronegative OH- ions. As the pH of the subphase water increases, the amount of OH- ions increases and so the amount of oxide coverage. Also, as the duration for which the substrate was inside subphase water increases, the chances of chemical reaction increases and so the amount of oxide coverage, but never complete or full. The attachment of Ni head groups on the oxide portion of the partially transformed Si substrate by deposition of LB film, essentially weaken the nearby Si-Si bonding, which then readily react with oxygen to form oxides. It is observed that depending upon the amount of attached Ni head groups and its distribution on the Si surface, it is possible to have complete oxide coverage through such reaction in air. The signature of which, from structural point of view, is only visible after subsequent deposition, i.e. after reorganization of molecules under subphase water [published in Chem. Phys. Lett. 500, 90 (2010)].
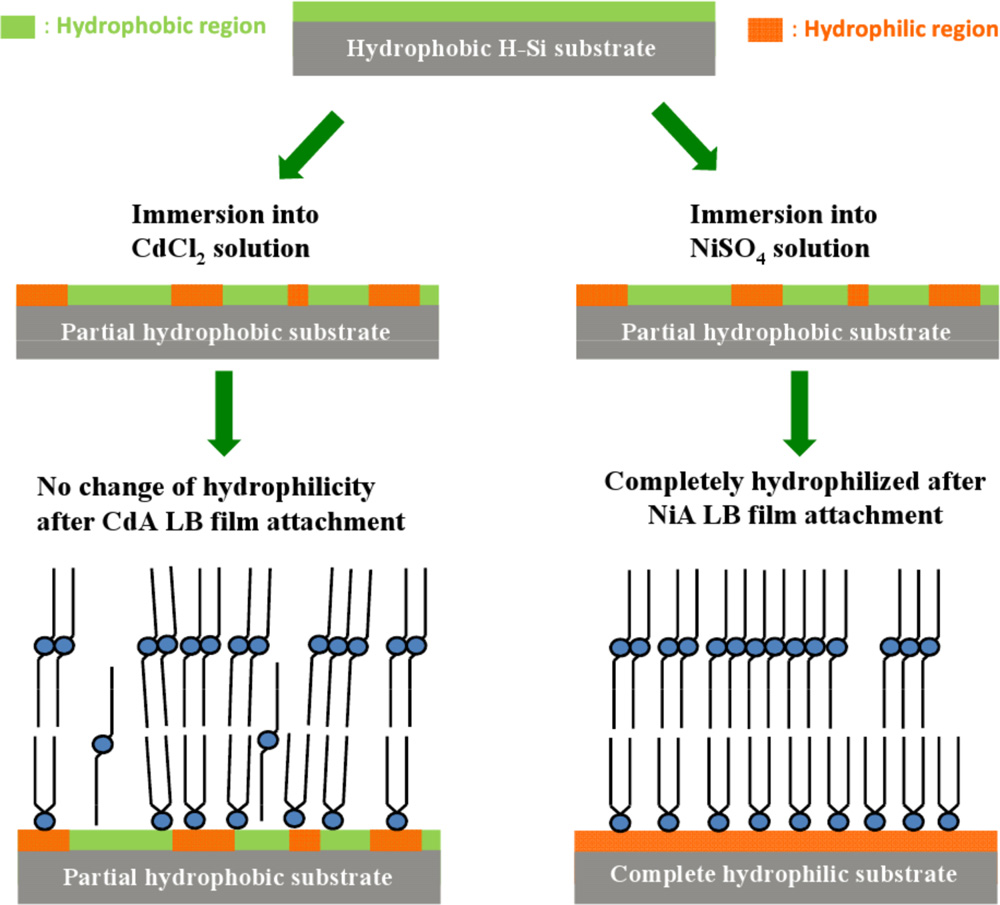 Hydrophobic to hydrophilic transition of HF-treated Si surface strongly depends upon the metal ions, which are present in the headgroups of the deposited Langmuir–Blodgett (LB) film. Structure of LB films studied by X-ray reflectivity technique and chemical analysis of LB film-substrate interfaces studied by X-ray photoelectron spectroscopy suggest that the partial transition or partial oxidation of the HF-treated Si surface takes place under the subphase water but further transition or oxidation is possible only in the presence of metal ions. Electrovalent and covalent natures of the metal ions tune this transition or oxidation. Ni ions, for which bonding with headgroups is electrovalent in nature, are favorable for such transition/oxidation and as a result, complete transition/oxidation takes place when nickel arachidate LB film is deposited. On the other hand, Cd ions, for which bonding with headgroups show covalent nature, is not favorable for such transition and cannot oxidize the underlying H-passivated Si substrate totally when cadmium arachidate LB film is deposited on such HF-treated Si surface. This ion-specific hydrophobic to hydrophilic transition is visualized by X-ray reflectivity, contact angle and X-ray photoelectron spectroscopy measurements [published in Mater. Chem. Phys. 134, 549 (2012)].
Hydrophobic to hydrophilic transition of HF-treated Si surface strongly depends upon the metal ions, which are present in the headgroups of the deposited Langmuir–Blodgett (LB) film. Structure of LB films studied by X-ray reflectivity technique and chemical analysis of LB film-substrate interfaces studied by X-ray photoelectron spectroscopy suggest that the partial transition or partial oxidation of the HF-treated Si surface takes place under the subphase water but further transition or oxidation is possible only in the presence of metal ions. Electrovalent and covalent natures of the metal ions tune this transition or oxidation. Ni ions, for which bonding with headgroups is electrovalent in nature, are favorable for such transition/oxidation and as a result, complete transition/oxidation takes place when nickel arachidate LB film is deposited. On the other hand, Cd ions, for which bonding with headgroups show covalent nature, is not favorable for such transition and cannot oxidize the underlying H-passivated Si substrate totally when cadmium arachidate LB film is deposited on such HF-treated Si surface. This ion-specific hydrophobic to hydrophilic transition is visualized by X-ray reflectivity, contact angle and X-ray photoelectron spectroscopy measurements [published in Mater. Chem. Phys. 134, 549 (2012)].
|

